An easy way to pick up marks in the GCSE Physics exam is by balancing nuclear equations. To do this, you need to apply the law of the conservation of mass, which says the total mass before a nuclear change takes place is equal to the total mass after the change has occurred.
This is fairly straightforward for natural decay, where one nucleus decays into a different nucleus with the release of either alpha or beta radiation.
Here is an example; the decay of americium-241 to form a “mystery atom” by the release of an alpha particle, as shown below.
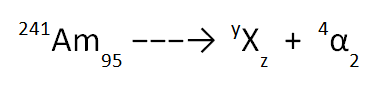
We can work out the numbers for our mystery atom by remembering that the numbers on the left of the equation must be the same as the totals of the numbers on the right (taking the top and bottom values separately).
If we look at the top numbers, we see 241 → y + 4. This means that the value of y must be 237.
Similarly, if we look at the bottom numbers, we see 95 → z + 2. This means that the value of z must be 93.
The final equation, written in full with the correct element symbol, is therefore as shown below.
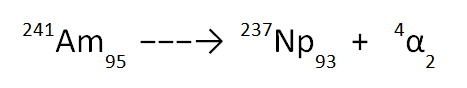
Note that you will NOT be required to remember the element symbol of the new nucleus; you only need to be able to balance the equation.
You must be especially careful when balancing a beta decay equation because the atomic number for a beta particle is -1. The equation shown below is correctly balanced but you should look carefully at this example to make sure that you understand WHY it is correctly balanced.
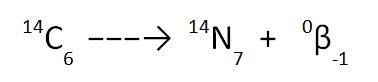
Things are a bit more complicated in the case of nuclear fission, where a nucleus captures a neutron and becomes unstable, splitting into two different “daughter” nuclei and releasing further neutrons. The method for balancing a fission equation is still the same but you must remember to include the masses of the incoming neutron and any free neutrons that are released.
Let’s look at the incomplete equation below and use our knowledge of the law of the conservation of mass to work out the missing values, p and q.
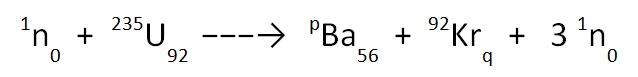
On the top row of the equation we have; 1 + 235 → p + 92 + (3 x 1). This gives a value of p equal to 141. Note that the large “3” acts to multiply everything that follows it; both the 1 at the top and the 0 at the bottom.
And on the bottom row we have; 0 + 92 → 56 + q + (3 x 0). This gives a value of q equal to 36.
The final equation is therefore as shown below.
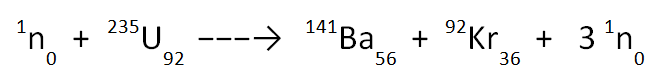
This is probably the highest level of challenge that you are likely to meet for GCSE so if you are happy with the example above then you should be able to answer anything the examination throws at you!

2 thoughts on “Balancing Nuclear Equations”